
As with many rules, there are exceptions, or violations. 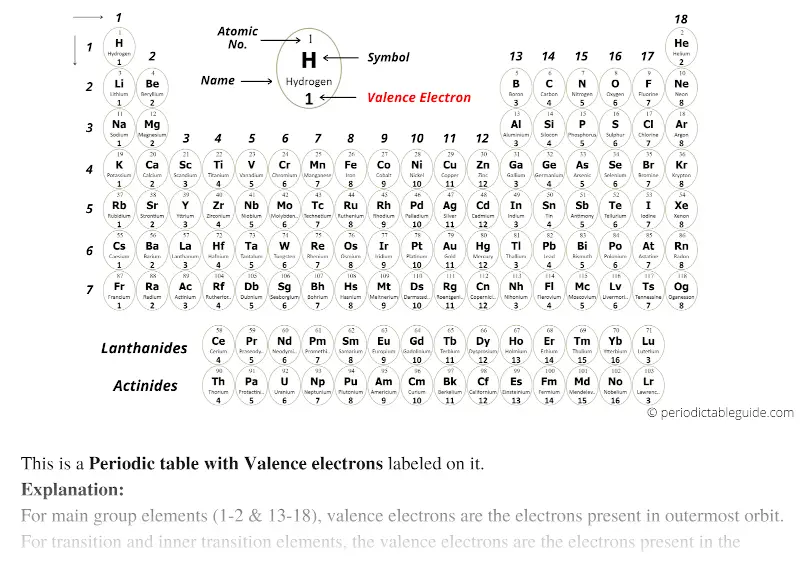
This does not mean that the octet rule is useless-quite the contrary. Always make sure all valence electrons are accounted for and each atom has an octet of electrons except for hydrogen (with two electrons).Īs important and useful as the octet rule is in chemical bonding, there are some well-known violations.This will not change the number of electrons on the terminal atoms.If the central atom has fewer electrons than an octet, use lone pairs from terminal atoms to form multiple (double or triple) bonds to the central atom to achieve an octet. 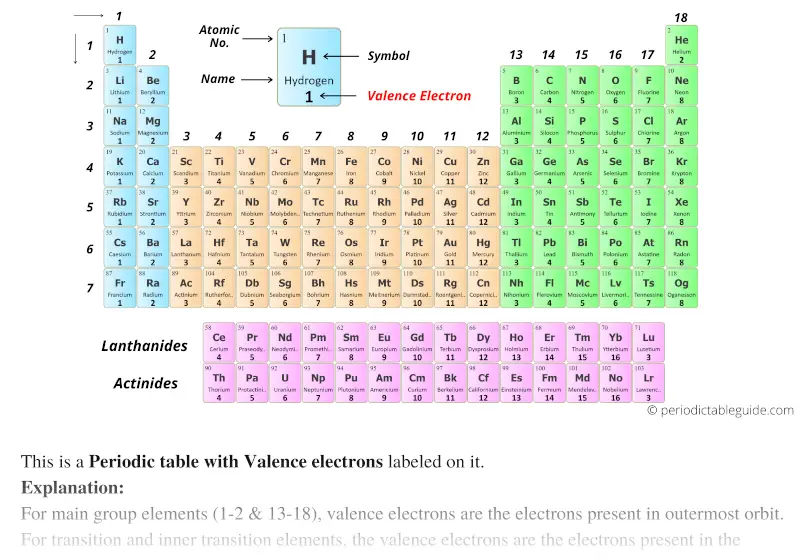
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
